0462
Parallel processes of neuro-vascular and neuro-cellular coupling1Emory University/Georgia Institute of Technology, Atlanta, GA, United States
Synopsis
Neural activity leads to cellular swelling along with the hemodynamic response from the vasculature. The latter process is the basis for the BOLD signal detected with fMRI; the former may underlie the changes observed with diffusion-weighted fMRI. Optical intrinsic signals can detect neuro-vascular activity (typically observed in in vivo studies of reflectance) and neuro-cellular swelling (observed mostly by transmittance in brain slices). We designed a novel miniature probe for in vivo transmittance studies in the rat brain and examined neuro-cellular coupling and neuro-vascular coupling in vivo to better understand the basis of the MRI techniques.
Purpose
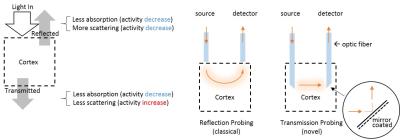
In vitro brain slice optical studies of light transmittance show that cells may swell during neural activity [1]. In the intact brain, however, these signals are relatively little studied compared to the hemodynamic response that is widely used to represent neuronal activity in neuroimaging. We designed a novel transmission-detection-mode probe to measure light transmittance in vivo in the rat brain. Transmitted light signals can separate absorption and scattering by the direction of their response to neuronal activity (figure 1), allowing signal from cell swelling to be isolated from the hemodynamic signals. Using simultaneous OIS and microelectrode recordings, we investigated the properties of neuro-cellular coupling and compared them to neurovascular coupling during spontaneous brain activity.Methods
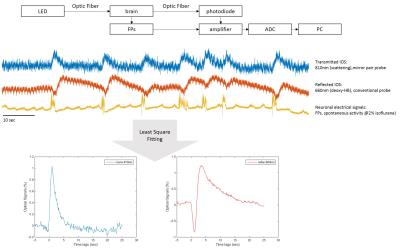
In vivo functional optical imaging studies detect reflected light. We compared signals from the newly-developed transmission mode (in cortex) to the conventional reflection detection mode (over the brain surface) and to recordings of neuronal electrical field potentials (FPs) in 9 male Sprague-Dawley rats. Small optical fibers (core diameter = 105 microns) with 45° sloped tips (mirror coated) were used for minimally invasive studies of light transmittance. The source-detector fiber pair were set 2mm apart. Signals were recorded during spontaneous burst-suppression activities under 2% isoflurane. The optical responses were examined for 7min for each session. Multiple sessions for each animal were conducted for different wavelengths, including isosbestic points at 525nm and 810nm (CBV contrast), and also 660nm (10-fold greater deoxy-HB absorption than oxy-HB; BOLD contrast). Both field potentials and optical signals were amplified in DC amplifiers and digitalized at 2400 Hz. The averaged response function for each session was calculated by least square fitting. Figure 2 illustrates schematically the experiments. Group data (mean+/-SEM) were used for comparison of response time courses across detection modes and wavelengths.Results
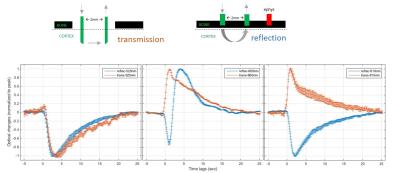
We compared conventional reflected signals with transmitted signals in somatosensory cortex (figure 3). Significantly, the transmission detection detected an increase in transmitted signals in response to bursts of field potentials at the longer wavelengths. The transmission increase coincides with previous in vitro brain slice studies of neuronal activity-dependent swelling dynamics, with a similar peak time of about 1 sec [1]. Meanwhile at the short wavelengths where absorption dominates, such as 525nm, similar time courses of light attenuation by hemoglobin absorption with a peak time of 4 sec for both detection modes of reflection and transmission were observed. A characteristic deoxygenation signal at 660nm (10-fold deoxy-HB absorption than oxy-HB) was observed in conventional reflection mode and exhibited an initial dip. The transmitted increases cannot be interpreted as a vascular response because the response is opposite in direction (signal would decrease in the case of hemoglobin absorption). This indicates that scattering is dominant. Furthermore, the signal latency is 1 sec, which is similar to the BOLD initial dip, but faster than the CBV or positive BOLD response (4 sec latency).Discussion/ Conclusion
We conducted a first in-vivo optical transmission mode study in the rat brain, and found evidence of transmission increases similar to those observed in brain slices due to activity-dependent neural cellular swelling. The cell swelling signal has a separate origin from the hemoglobin signals arising from neurovascular coupling but occurs on a similar time scale. The transmission signal exhibits a faster latency (1-sec peak time) than vascular responses (4-sec in CBV), similar to initial dip of oxygenation. The fast latency in neuro-cellular coupling of optical signals is also consistent with diffusion-based fMRI findings [2]. In conclusion, the neuro-cellular coupling may be a significant bio-marker for neural activity in vivo and provide a complementary contrast to neuro-vascular coupling.Acknowledgements
•NIH, R01 NS078095References
1. Pal, I., et al., Neuronal and astroglial correlates underlying spatiotemporal intrinsic optical signal in the rat hippocampal slice. PLoS One, 2013. 8(3): p. e57694.
2. Le Bihan, D. and M. Iima, Diffusion Magnetic Resonance Imaging: What Water Tells Us about Biological Tissues. PLoS Biol, 2015. 13(7): p. e1002203.
Figures