0372
7T Diffusion Tensor Imaging of High Mechanical Stress Achilles Tendon-to-Bone InterfaceSimone Angela Winkler1, Lara Kuntz1,2, Christoph Leuze1, Hossein Nejadnik1, Laura J Pisani1, and Heike Daldrup-Link1
1Dept of Radiology, Stanford University, Stanford, CA, United States, 2Clinics of Orthopedics and Chair of Cellular Biophysics, Technische Universitaet Muenchen, Munich, Germany
Synopsis
We present imaging analysis using MRI of the Achilles tendon-to-bone interface (“enthesis”) to investigate on the four distinct zones responsible for mechanical force transfer. Conventional T1- and T2-weighted imaging in humans yields very low MR signal. We therefore present T1-weighted FLASH 3D imaging at ultra high-field (7T), using a porcine sample from a minipig leg. We were able to identify the four zones (fibrous connective tissue, uncalcified fibrocartilage, calcified fibrocartilage, and bone) in both T1 and diffusion tensor imaging (DTI).
Introduction
Tendons act as a mechanical element for force transfer between different parts of the skeleton. The attachment site between tendon and bone is commonly referred to as “enthesis” and is the site for high concentration of mechanical stress and as such often vulnerable to sports injuries of acute or overuse type. In addition, rheumatic conditions often target enthuses with inflammation. Entheses are of particular importance to 1) surgeons who reattach tendons, and 2) tissue engineering applications, in which the interface is engineered and added artificially. Literature has shown that four distinctive zones can be found in entheses: fibrous connective tissue, uncalcified fibrocartilage, calcified fibrocartilage, and bone. In this study, we investigate the enthesis using high-field MRI techniques at 7 Tesla, using a protocol of 1) T1 FLASH imaging and 2) Diffusion Tensor Imaging (DTI). Normally, T1-weighted imaging of tendon and enthesis yields little MR signal. We hypothesize that studying the interface in a young pig and with the benefit of intrinsic increased SNR as well as better structural delineation at high field strength offer increased information on the anatomical composition of the four enthesis zones.Methods
Sample preparation: A porcine sample of the Achilles tendon-bone interface was prepared from a minipig leg, with approximate dimensions of 13 mm x 13 mm x 20 mm. The sample was fixed using formalin for 24 hours and subsequently washed in phosphate buffered saline for 4 hours. The sample was inserted in fomblin to eliminate MR signal from the background. Hardware setup: We placed the sample in a birdcage transmit-receive coil at room temperature and acquired images with an Agilent/Bruker 7T small-bore MRI system. Imaging sequence setup: We acquired coronal T1 FLASH images using 3D acquisition to maximize signal-to-noise ratio (SNR) and TE/TR/FA=20ms/58.6ms/22deg with a field of view (FOV) of 24mmx14mmx14mm, at an isotropic resolution of 100um, leading to an acquisition time of 21min. We then acquired coronal DTI images with 3D-EPI sequence with TE/TR =26.4ms/500ms, 100um isotropic resolution, b=1000s/mm2, 2 averages, 2 readout segments, 120 diffusion directions, 20 b0 images, gradient duration and separation of 3ms and 10ms, respectively. The total scan time was 10h53min. Data reconstruction and evaluation: T1 images were investigated for high-intensity regions and anatomical information to delineate the four enthesis zones. Diffusion tensors were calculated and visualized using the FSL toolkit [4]. Diffusion data was downsampled to 200um to increase voxel-wise SNR before diffusion tensor estimation. We compared the zonal composition to a micro computed tomography image and human histology.Results
Fig. 1 shows the T1 FLASH image and depicts the anatomical composition and layering of the tendon-bone interface in the heel of a young pig. We can identify four distinct enthesis zones and their dimensions: (1) dense fibrous connective tissue (~0.3mm), (2) uncalcified fibrocartilage (~0.8mm), (3) calcified fibrocartilage (~0.3mm), and (4) bone. These measurements roughly agree in proportion with the findings for histology of human samples in [1]. These zones are not conventionally delineable with T1 imaging at 3T and in human tissue. Fig. 3 shows the primary eigenvector of the diffusion tensor, whose color depicts the direction of the vector. We can clearly delineate the four compositional zones of the tendon-bone interface and assign orthogonal diffusion directions for each of the zones. The diffusivity in dense fibrous connective tissue occurs predominantly along the longitudinal direction of the tendon fibers, while diffusivity in uncalcified fibrocartilage roughly follows a right-left direction with respect to the foot. The calcified bone layer shows a larger likelihood of water diffusion in the radial direction with respect to the bone center, whereas in the bone itself no distinct diffusion direction is observed.Discussion
Future work may include increased resolution for the T1 FLASH acquisition to 50um to reveal further details on the enthesis zone composition, as well as UTE T2 imaging in humans at 7T [5] using ultra-short TE times to reveal MR signal from bone.Conclusions
This study provides confirmation for the use of T1 imaging and DTI in Achilles tendon-bone interface using histology in a young pig. Comparison to micro computed tomography imaging confirms proportional anatomical composition and layering.Acknowledgements
No acknowledgement found.References
[1] Benjamin M, J Anat, 208:471-90 (2006). [2,3] Insert ISMRM paper and PhD thesis. [4] Smith SM, et al. NeuroImage, 23(S1):208-19 (2004). [5] Han M et al., Invest Radiol 49(5):339-45.Figures
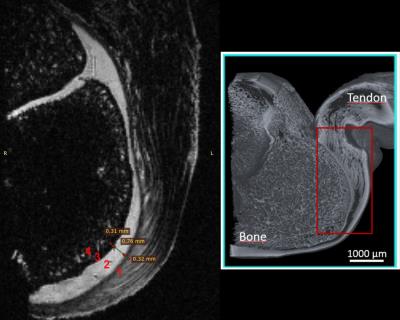
Figure
1: left: T1 FLASH image (FOV=24mmx14mmx15mm, resolution=100um isotropic,
TE/TR/FA=20ms/58.6ms/22deg). Note the four enthesis zones and their dimensions:
(1) dense fibrous connective tissue (~0.3mm), (2) uncalcified fibrocartilage
(~0.8mm), (3) calcified fibrocartilage (~0.3mm), and (4) bone; right: micro
computed tomography image of human Achilles tendon-bone interface delineating
the four enethesis zones.
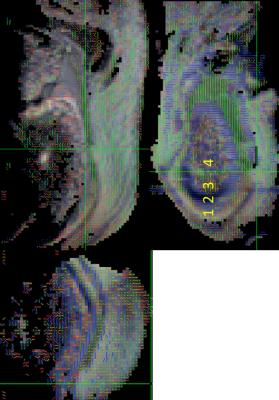
Figure
2: DTI images in coronal, sagittal, and axial orientations: We can clearly
delineate the four enthesis zones, which differ in diffusion direction. (1)
dense fibrous connective tissue, (2) uncalcified fibrocartilage, (3) calcified
fibrocartilage, and (4) bone.