1553
Imaging of Stroke in Rats using a Clinical Scanner and an Inductively Coupled Specially Designed Receiver Coil1Systems Neuroscience Lab, Centro de Investigación Médica Aplicada, Pamplona, Spain, 2Neos Biotec, Pamplona, Spain, 3Radiology, Clínica Universidad de Navarra, Pamplona, Spain
Synopsis
Imaging of small laboratory animals in clinical MRI scanners is feasible but challenging. Compared with dedicated preclinical systems, clinical scanners have relatively low main field (1.5 – 3.0 T) and gradient strength (40 – 60 mT/m). This work explores the use of wireless inductively coupled coils combined with adapted pulse sequences to overcome these two drawbacks, with a special emphasis on the optimization of the coil passive detuning circuit for this application. The images of rat brain stroke obtained show a substantial increase in SNR compared to clinical coils, and the absence of wires makes the animal preparation workflow straightforward.
Introduction
In-vivo preclinical MRI is an important tool in translational research. Dedicated high-field preclinical scanners provide high-quality images but are expensive and not readily accessible. Clinical scanners, conversely, have become widely available and offer an alternative to researchers carrying out animal studies1. Although the feasibility of performing small-animal imaging in clinical scanners has been demonstrated2, the lower field and gradient strengths present challenges and limitations. The lower field reduces the SNR; however, SNR could be increased by the selection of smaller receiver coils. The goal of this study was to evaluate the use of an inductively coupled receiver coil (ICC) of reduced dimensions to image small animals in a 3T-scanner. This type of coil3 has interesting features for this application: (1) minimum number of components; (2) interoperability with scanners from different manufacturers; (3) absence of cables, which makes the coil inherently balanced, minimizing any parasitic coupling to the surrounding tissue, and avoiding the associated SNR losses. In this work, the performance of the coil was evaluated in a rat model of stroke.Methods
RF Coil: The ICC consisted of a wireless, inductively coupled resonant loop4, tuned to the Larmor frequency, and used in combination with a sniffer coil that transferred the signal to the scanner receiver. To maintain the homogeneity of the B1+ transmit field, the wireless loop was fitted with a passive detuning circuit. Different circuit designs were studied: antiparallel limiter diode pair, PIN diode biased by RF rectifying circuit, Enhancement Mode Gallium Nitride field effect transistor and Schottky diode pair. The effect of the detuning tank circuit in the Q spoiling was also analyzed and tested, leading to the conclusion that this tank circuit is not particularly useful for our passive detuning application, due to the non-linear behavior of the RF switches, which is power-level dependent.
Animals: Five adult male Sprague-Dawley rats were used (250-300 g). Animal care and procedures were approved by the ethics committee of the University of Navarra.
Infarct induction: Rats were anesthetized and a longitudinal incision along the midline scalp was performed. Then, a dose of 150mg/kg of Rose Bengal was injected intraperitoneally. After five minutes, a cold light beam was applied for 30 minutes by direct contact to the skull. After that, the wound was cleaned and sutured.
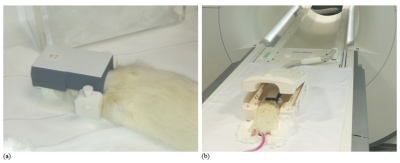
Scanning protocol: Rats were scanned under anesthesia one day after infarct induction. In addition, one animal was re-scanned after one week. The rat was placed in prone position on a custom-built support, which was introduced inside the clinical wrist-coil. The ICC was positioned over the rat head (Fig. 1). Imaging was performed on a 3T-Siemens Skyra (45 mT/m gradient strength) using the body-coil for RF transmission. The scanning session included T2 and T1-weighted sequences (see parameters in Table 1), run in coronal orientation.
SNR measurements: An ROI was drawn in the rat cortex (Fig. 2), contralateral to the infarcted side. SNR was calculated as the ratio of the mean ROI signal to the standard deviation of the background signal.
Histology: Brain fixation was achieved by transcardial perfusion with paraformaldehyde(PFA) under anesthesia. Brain was extracted, maintained in PFA for 24 hours and changed to PBS-sacarose for at least 24 hours before tissue processing. 40 mm slices were cut and stained with thionin.
Results and Discussion
The conditions of our application (small coils, clinical scanner, 3T) made the coil passive detuning challenging for low-power RF pulses. Unlike other applications, where power handling capability of the detuning circuit is crucial, the dimensions of our coil made it pick up relatively little energy from the body-coil, simplifying the thermal requirements of the detuning circuit. However, it made the switching of the passive detuning circuit very difficult for low flip-angle RF pulses. This was noticeable in the T1-weighted images, where most of the tested detuning circuits produced artifacts. The final design of the circuit (Schottky diode pair without detuning trap) solved this issue and allowed acquisition of high quality T2 and T1-weighted images of the rat brain (Figs. 2 and 3). The SNR of the images acquired with the ICC was significantly higher than the SNR of those acquired with the wrist-coil alone, by a factor of 6 to 7 (Table 2). The high-quality images obtained with the ICC allowed a clear visualization of the infarct, both acute and chronic (Fig. 3), providing excellent correlation with histology.Conclusion
This work demonstrates the feasibility of using wireless ICCs to image small animals in clinical scanners. This technique can offer an alternative to research groups which do not have access to a preclinical imaging system.Acknowledgements
Government of Navarra Grant 0011-1365-2017-000106.References
1. Bilgen M. Feasibility and merits of performing preclinical imaging on clinical radiology and nuclear medicine systems. Int J Mol Imaging ID 923823 (2013).
2. Brockmann M, et al. Current issues and perspectives in small rodent magnetic resonance imaging using clinical MRI scanners. Methods 43:79-87 (2007).
3. Graft H., et al. Inductively coupled RF coils for examinations of small animals and objects in standard whole-body MR scanners. Med Phys 30: 1241-6 (2003).
4. M. D. Schnall, C. Barlow, V. Harihara Subramanian, J. S. Leigh Jr. Wireless Implanted Magnetic Resonance Probes for in Vivo NMR. J Magn Reson 68, 161-167 (1986).
Figures